Nonclinical Toxicology Stendra
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis and Impairment of Fertility
Carcinogenesis
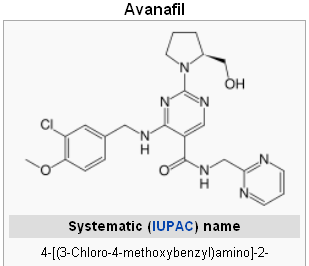
Avanafil was not carcinogenic to CD-1 mice when administered daily at doses of 100, 200, or 600 mg/kg/day orally by gavage for at least 98 weeks (approximately 11 times the MRHD on an AUC basis) so they can Sprague Dawley rats when administered daily at doses of 100, 300, or 1000 mg/kg/day orally by gavage not less than 100 weeks (approximately 8 times for males and 34 times for females above the MRHD with an AUC basis).
Mutagenesis
Avanafil hasn't been genotoxic inside a selection of tests. Avanafil had not been
mutagenic in
Impairment of love and fertility
Within a rat fertility and early embryonic development study administered 100, 300, or 1000 mg/kg/day for 28 days ahead of paring and continued until euthanasia for males, and 2 weeks previous to pairing to gestation day 7 for females, a decrease in fertility, no or reduced sperm motility, altered estrous cycles, and an increased percentage of abnormal sperm (broken sperm with detached heads) occurred at exposures in males approximately 11 times the human beings exposure with a dose of 200 mg. The altered sperm effects were reversible on the end of a 9-week drug-free period. Systemic exposure for the NOAEL (300 mg/kg/day) was similar to a person's AUC on the MRHD of 200 mg.
Animal Toxicology and/or Pharmacology
Repeated oral administration of avanafil in multiple species triggered signs of centrally-mediated toxicity including ataxia, tremor, convulsion, hypoactivity, recumbency, and/or prostration at doses creating exposures approximately 5-8 times the MRHD determined by Cmax and 8-30 times the MRHD based on AUC.